The Meals and Drug Administration (FDA) has granted 510(ok) clearance for the Provisio SLT IVUS System, which incorporates a help crossing catheter that integrates intravascular ultrasound, enabling clinicians to evaluate blood vessel lumen and acquire vessel measurements on the similar time via Sonic Lumen Tomography (SLT).
Provisio Medical , the producer of the Proviso SLT IVUS System, mentioned the Provisio SLT IVUS System’s inclusion of the SLT IVUS Assist Crossing Catheter offers twin performance in interventional procedures with an ultrasound transducer array on the distal finish of the catheter.
Via the usage of Sonic Lumen Tomography (SLT), the newly FDA-cleared Provisio SLT IVUS System offers clinicians with simultaneous real-time visualization and measurement of vessel lumen throughout interventional procedures for sufferers with peripheral vascular illness. (Picture courtesy of Provisio Medical.)
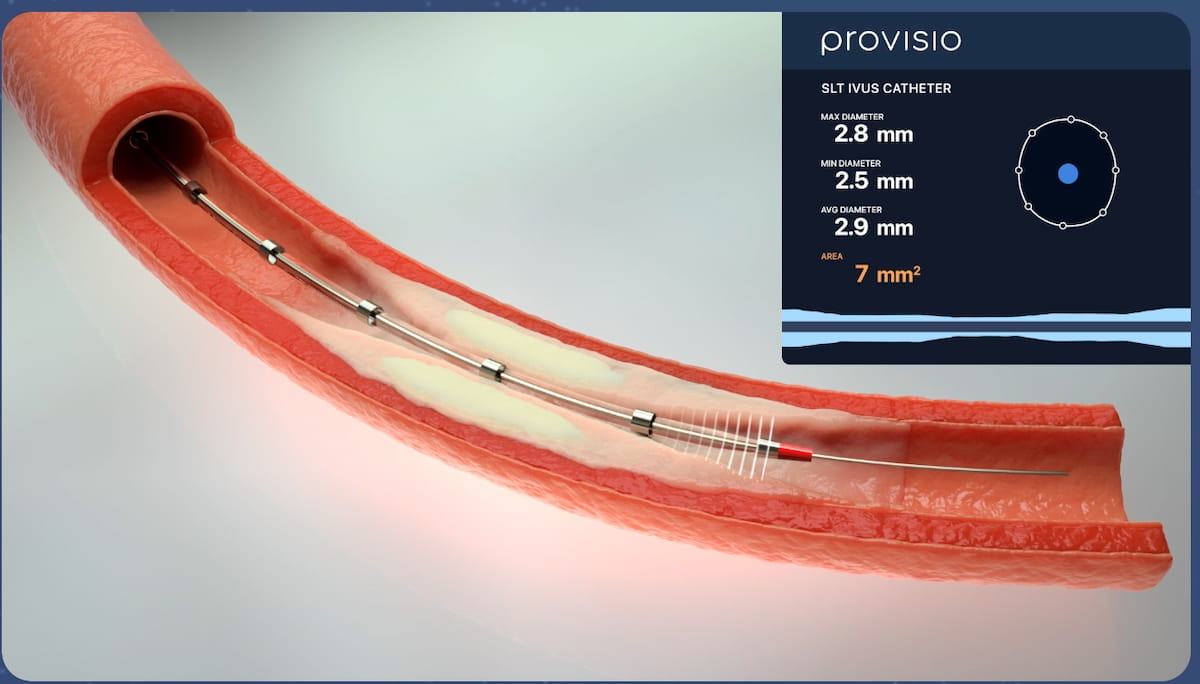
Incorporating intravascular ultrasound functionality right into a front-line help crossing catheter might assist simplify workflows, cut back iodinated distinction media (ICM) and cut back radiation publicity for sufferers being handled for peripheral vascular illness (PVD), in accordance with Provisio Medical.
“Medical outcomes in peripheral vascular illness have persistently been proven to profit from correct intravascular measurements, but adoption has been restricted by the extra process time and coaching required to interpret photographs” famous S. Eric Ryan, M.D., the chief govt officer of Provisio Medical. “Due to the ease-of-use of SLT IVUS, which will be included extra effectively within the peripheral vascular workflow, we imagine there may be the potential of elevated adoption and subsequently improved outcomes for a lot of extra sufferers with probably devastating peripheral vascular illness.”

